#DIFFERENCE BETWEEN ISO 9001 AND ISO 13485 ISO#
If the organization certified by ISO 27001 standard, it means the organizations can manage the information security risks by implementing the standard along with other supporting standard ISO 27002. Now coming to ISO 27001, it is an International standard which focuses on the useful information security management system of the organization by implementing the standard. If the organization has achieved an ISO 9001 certification, it means there is a successful demonstration of the process which involves the product or service of the operation, customer focus, development and design of services and products, infrastructure, input and output designs, and how the process managed externally. One of the great combinations of the certification which has gained more popularity is ISO 9001, quality management system, and ISO 27001, information security management system.Īs discussed earlier ISO 9001 is a quality management system which specifies the requirement for an industry or organization to demonstrate its capability to provide the entire product and services consistently to meet the regulatory and customer needs. Let us know more about ISO 9001 vs ISO 27001Įvery organization works to meet a customer and legal requirements, so it is becoming more common to the organizations to maintain and obtain the multiple ISO certifications. Not only that also includes provisions how product manufactured, but quality also has to test, and how the information security.ĭepending upon the process and scope of the organization, ISO has published more than 22000 + of standards throughout the globe.Īmong which ISO 9001 is one of the first and generic standards which can apply to all the organization which is looking for customer satisfaction and the process improvement.Īnd another standard ISO 27001 specifies the requirement for an information security management system it provides a Framework for procedures and policies which include all the physical, legal and technical controls involved in the organization’s information management processes. The main aim of ISO is to publish the standards, and it defines the norms and rules to overcome the risks for all the products that people use.
#DIFFERENCE BETWEEN ISO 9001 AND ISO 13485 HOW TO#
Some key differences between 905 include standards for customer feedback, monitoring product performance, and how to address a non-conforming product.ISO 9001 vs ISO 27001 is an acronym for an international organization for standardization which established in the year 1947 and the headquarters in Geneva, Switzerland. ISO 13485 creates a more granular definition of the types of improvement activities device manufacturers need to ensure products are safe and effective. ISO 9001 takes a process-driven approach to continual improvement. 134 emphasize employee competency and infrastructure for quality.Both 905 use Deming cycles (Plan-Do-Check-Act).A focus on the realization of quality products through understanding the customer.Risk mitigation and assessment is a significant focus in both standards.The standard’s role in helping organizations achieve a quality management system.There are significant similarities between the two standards, including: Medical devices – Quality management systems – Requirements for regulatory purposes represent the requirements that the medical device manufacturers must incorporate into their management systems, ISO 13485 documents kit to comply with Medical Devices manufacturing QMS. It places a more significant focus on regulatory compliance and offers less flexibility in the organizational process. ISO 13485 is a comprehensive management system specifically for the manufacture of medical devices. ISO 9001 documentspackage is designed based on the best quality manufacturing practices and processes approach. ISO 9001 is the international standard which provides specifications for a quality management system which can be applied at any organization regardless of industry, product or service, or company size.

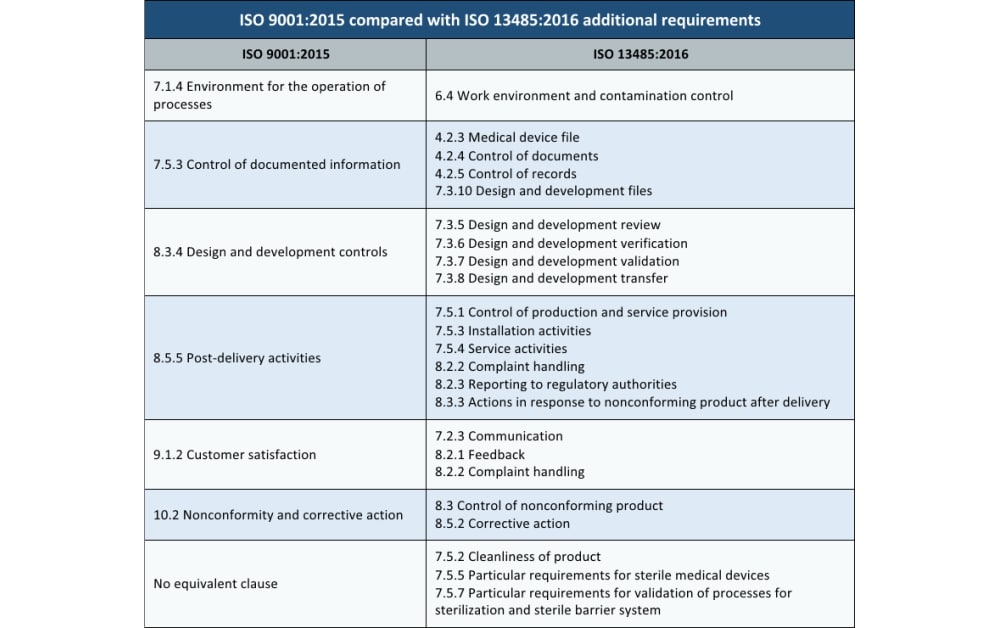
The primary difference between ISO 13485 and ISO 9001 for medical devices is the scope of these quality standards. ISO 13485 and ISO 9001 for Medical Devices: How They Differ By understanding the differences between these two standards, you learn where device manufacturers need to raise the bar on quality. What many medical device manufacturers fail to realize, however, is that comparing ISO 9001 and ISO 13485 is a valuable exercise.

ISO 13485 is a quality system for the medical device industry, and it effectively covers ISO 9001 with some additional requirements.
